70% of advisors under the age of 40 use separately managed accounts; 67% use ESG products;
25% of advisors plan to increase their engagement with wholesalers in 2021, showing the changing dynamics in the advisor-wholesaler relationship
PR Newswire
NEW YORK, April 19, 2021 /PRNewswire/ — Broadridge Financial Solutions, Inc. (NYSE: BR), a global Fintech leader, today released results from an annual survey of 400 financial advisors, revealing that a majority of advisors across channels use ESG products, citing direct interest from investors. The study also shows how the advisor-wholesaler relationship is evolving post-pandemic.
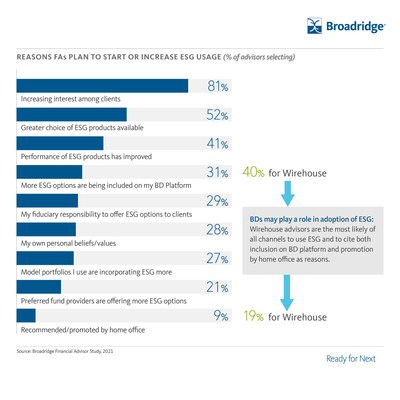
Sixty-one percent of advisors use ESG products, and usage is even higher among female advisors (71%) and advisors under 40 (67%). Of advisors who use ESG products, 81% plan to increase their usage over the next two years as investors actively seek out these products. Wirehouse advisors are the most likely of all channels to use ESG products and to cite both inclusion on their broker-dealer platform and promotion by their home offices as reasons for doing so.
Sixty-two percent of financial advisors use separately managed accounts (SMAs), and that number is even higher among advisors under 40 (70%). Fifty-six percent of advisors who use SMAs plan to increase their usage over the next two years.
As advisors explore new investment vehicles such as ESG and SMAs, half of advisors (51%) are now also using private funds. Eighty-one percent of advisors who use private funds also use SMAs.
“Now more than ever, investors are speaking up about the types of products they want to invest in, whether it be ESG, private markets or other emerging asset classes. As advisors become more focused on providing the best client-centric experience and are optimistic about the year to come, they are looking to be equipped with the right tools and products to service their clients and investments,” said Matthew Schiffman, Principal of Distribution Insight at Broadridge Financial Solutions. “Asset managers are primed to step up and address the demand for these products.”
The “New Normal” For Advisor-Wholesaler Relationships
On average, advisors maintain 48% of their AUM with their top-five asset management firms. However, 28% of advisors are very open to adding new asset managers, identifying an opportunity for asset managers to uncover new ways to showcase their value, win over prospects and retain AUM.
A quarter of advisors (25%) plan to engage with external wholesalers more in 2021 and 70% of advisors say that an external wholesaler is one of their top-three most preferred methods of engaging with an asset manager.
“Forced into a fully remote environment just over a year ago, many advisors dramatically accelerated their digital journey to maintain and grow their business. Today’s advisors are actively focused on business development and client-facing activities, and are increasingly using model portfolios as part of their practice to allow for increased efficiency in a post-COVID world,” added Schiffman. “Advisors were not able to engage with wholesalers in the same way they did before the pandemic and were initially hesitant to engage virtually. Now, as the COVID-19 vaccine rollouts progress and we may see a return to normal sooner than expected, advisors are eager to re-engage with wholesalers and are receptive to new relationships.”
Advisors continue to feel positive about conditions for success today and in the future, with the study showing that 77% are favorable (somewhat or very) about growing and scaling their practice today, and 67% expect conditions to be better three years from now. Further, 66% of advisors are confident (somewhat or very) in acquiring new clients today and 56% expect to see improvements three years from now.
View Broadridge’s 2021 report The Evolving Advice Business Model.
Methodology
This Broadridge survey was conducted by 8 Acre Perspective to assess the world of financial advice and guidance. A total of 400 financial advisors across wire, regional, IBD and RIA channels completed the survey, which was fielded in January and February 2021. For further details on survey methodology, please contact a Broadridge media representative.
About Broadridge
Broadridge Financial Solutions (NYSE: BR), a global Fintech leader with over $4.5 billion in revenues, provides the critical infrastructure that powers investing, corporate governance and communications to enable better financial lives. We deliver technology-driven solutions to banks, broker-dealers, asset and wealth managers and public companies. Broadridge’s infrastructure serves as a global communications hub enabling corporate governance by linking thousands of public companies and mutual funds to tens of millions of individual and institutional investors around the world. In addition, Broadridge’s technology and operations platforms underpin the daily trading of on average more than U.S. $10 trillion of equities, fixed income and other securities globally. A certified Great Place to Work®, Broadridge is a part of the S&P 500® Index, employing over 12,000 associates in 17 countries. For more information about us and what we can do for you, please visit www.broadridge.com.
Media Contacts:
Matthew Luongo
Prosek Partners
+1 646-818-9279
[email protected]
Linda Namias
Broadridge Financial Solutions
+1 631-254-7711
[email protected]
![]() View original content to download multimedia:http://www.prnewswire.com/news-releases/esg-and-sma-usage-primed-to-increase-as-financial-advisors-service-next-generation-investor-demand-new-broadridge-study-finds-301270660.html
View original content to download multimedia:http://www.prnewswire.com/news-releases/esg-and-sma-usage-primed-to-increase-as-financial-advisors-service-next-generation-investor-demand-new-broadridge-study-finds-301270660.html
SOURCE Broadridge Financial Solutions, Inc.