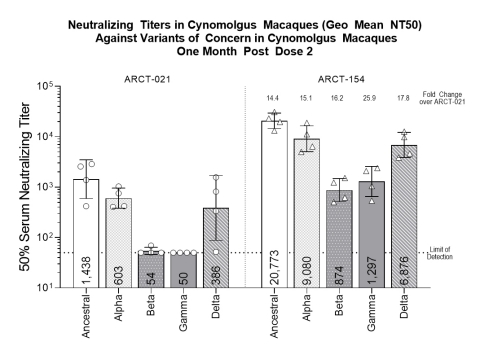
Everbridge Teams with The Associated Press (AP) to Integrate Global News Report into Market-Leading Critical Event Management (CEM) Platform
- Everbridge expands its market-leading risk intelligence offering to over 25,000 data sources, helping businesses, governments, and healthcare organizations correlate the things they care about (employees, citizens, facilities, supply chain, IT infrastructure) with real-time risks, in order to mitigate impact and accelerate response
- By adding AP breaking and geopolitical news into its Critical Event Management platform, Everbridge provides its customers with enhanced contextualization of risk data leveraging the reporting, speed and deep expertise of AP journalists in 250 locations worldwide
BURLINGTON, Mass.–(BUSINESS WIRE)–Everbridge, Inc. (NASDAQ:EVBG), the global leader in critical event management (CEM), and The Associated Press (AP) today announced a collaboration that will incorporate AP’s global news reports into the Everbridge Critical Event Management (CEM) platform. The collaboration will provide Everbridge customers with AP’s deep subject-matter expertise and extensive contextualization of risk data from critical events happening across the globe.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210809005689/en/

Everbridge Teams with The Associated Press (AP) to Integrate Global News Reports into Market-Leading Critical Event Management (CEM) Platform (Graphic: Business Wire)
Through machine learning and AI-enabled incident collection, Everbridge combines more than 25,000 of the most trustworthy data sources with an experienced team of analysts, creating the industry’s leading source of verified data and hyper local threat intelligence.
An independent global news organization with journalists in 250 locations in 100 countries, AP journalism is seen by more than half the world’s population each day through text stories, photos, videos, and live video coverage. For more than 175 years, AP journalists have kept the world informed of the biggest stories – from the COVID-19 pandemic to presidential elections to natural disasters.
Sourcing information from AP will allow Everbridge to provide enterprises and governments with one of the world’s top news feeds, curated along with thousands of additional risk sources. Leveraging comprehensive risk intelligence and contextualized situational analysis remains critical to how organizations effectively achieve enterprise resilience across digital and physical domains.
“We are excited to work with Everbridge to further expand the impact of AP’s geopolitical news coverage and broaden the number of organizations benefiting from the expertise of AP’s on-the-ground journalists around the world,” said Tom Januszewski, AP vice president of global business development. “AP’s journalism is trusted, reliable, accurate and fast, and we are pleased it will be used to help Everbridge clients react faster to critical events.”
Everbridge curates threat intelligence from risk data feeds monitoring weather, wildfires, floods, supply chain disruptions, man-made violence, terror threats and cyberattacks, among hundreds of other categories. Everbridge’s comprehensive, configurable risk monitoring capabilities deliver actionable information that helps reduce risk wherever people live, work, or travel. This intelligence helps to correlate, for example, the proximity of corporate assets to potential locations that may be subject to elevated risk. Risk visibility allows corporate security teams to easily determine required additional action to protect employees and assets around the world.
“Regarded as a top source for news from all corners of the world, AP provides extensive contextual content on real-time world events,” said Karl Kotalik, VP of global risk intelligence solutions. “By combining in-depth news reporting and analysis with risk event correlation, Everbridge provides our customers with the most comprehensive context to make informed decisions to manage and respond to risk.”
Everbridge’s end-to-end CEM platform digitally transforms how organizations manage and respond to critical events, helping organizations safeguard and enable revenue while reducing expenses. CEM can be deployed rapidly on a modular basis supporting hundreds of positive-ROI use cases including for people and life safety, operations and business continuity, supply chain risk, IoT and the smart enterprise, and IT incidents.
Everbridge customers include some of the largest firms and leaders in their respective industries including Fortune 1000 businesses such as Bristol Myers Squibb, Cisco, CVS Health, Goldman Sachs, Lowe’s, Tiffany & Co., chemical giant Dow, telecom consumer electronics company Nokia, as well as some of Silicon Valley’s leading tech giants, global e-commerce firms, oil and natural gas providers, hotel and hospitality chains, automotive, aerospace and defense technology, air travel, and major car rental firms.
Customers can also utilize Everbridge to share intelligence with other organizations and to coordinate response through the CEM platform, either publicly or privately. Everbridge’s support teams have established a multitude of private networks among law enforcement, hospitals, airports, universities and enterprises around the world so they can share critical event-related updates.
About AP
The Associated Press is an independent global news organization dedicated to factual reporting. Founded in 1846, AP today remains the most trusted source of fast, accurate, unbiased news in all formats and the essential provider of the technology and services vital to the news business. More than half the world’s population sees AP journalism every day. Online: www.ap.org
About Everbridge
Everbridge, Inc. (NASDAQ:EVBG) is a global software company that provides enterprise software applications that automate and accelerate organizations’ operational response to critical events in order to Keep People Safe and Organizations Running™. During public safety threats such as active shooter situations, terrorist attacks or severe weather conditions, as well as critical business events including IT outages, cyber-attacks or other incidents such as product recalls or supply-chain interruptions, over 5,700 global customers rely on the Company’s Critical Event Management Platform to quickly and reliably aggregate and assess threat data, locate people at risk and responders able to assist, automate the execution of pre-defined communications processes through the secure delivery to over 100 different communication modalities, and track progress on executing response plans. Everbridge serves 8 of the 10 largest U.S. cities, 9 of the 10 largest U.S.-based investment banks, 47 of the 50 busiest North American airports, 9 of the 10 largest global consulting firms, 8 of the 10 largest global automakers, 9 of the 10 largest U.S.-based health care providers, and 7 of the 10 largest technology companies in the world. Everbridge is based in Boston with additional offices in 25 cities around the globe. For more information visit www.everbridge.com.
Cautionary Language Concerning Forward-Looking Statements
This press release contains “forward-looking statements” within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including but not limited to, statements regarding the anticipated opportunity and trends for growth in our critical communications and enterprise safety applications and our overall business, our market opportunity, our expectations regarding sales of our products, our goal to maintain market leadership and extend the markets in which we compete for customers, and anticipated impact on financial results. These forward-looking statements are made as of the date of this press release and were based on current expectations, estimates, forecasts and projections as well as the beliefs and assumptions of management. Words such as “expect,” “anticipate,” “should,” “believe,” “target,” “project,” “goals,” “estimate,” “potential,” “predict,” “may,” “will,” “could,” “intend,” variations of these terms or the negative of these terms and similar expressions are intended to identify these forward-looking statements. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond our control. Our actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to: the ability of our products and services to perform as intended and meet our customers’ expectations; our ability to successfully integrate businesses and assets that we may acquire; our ability to attract new customers and retain and increase sales to existing customers; our ability to increase sales of our Mass Notification application and/or ability to increase sales of our other applications; developments in the market for targeted and contextually relevant critical communications or the associated regulatory environment; our estimates of market opportunity and forecasts of market growth may prove to be inaccurate; we have not been profitable on a consistent basis historically and may not achieve or maintain profitability in the future; the lengthy and unpredictable sales cycles for new customers; nature of our business exposes us to inherent liability risks; our ability to attract, integrate and retain qualified personnel; our ability to maintain successful relationships with our channel partners and technology partners; our ability to manage our growth effectively; our ability to respond to competitive pressures; potential liability related to privacy and security of personally identifiable information; our ability to protect our intellectual property rights, and the other risks detailed in our risk factors discussed in filings with the U.S. Securities and Exchange Commission (“SEC”), including but not limited to our Annual Report on Form 10-K for the year ended December 31, 2020 filed with the SEC on February 26, 2021. The forward-looking statements included in this press release represent our views as of the date of this press release. We undertake no intention or obligation to update or revise any forward-looking statements, whether as a result of new information, future events or otherwise. These forward-looking statements should not be relied upon as representing our views as of any date subsequent to the date of this press release.
All Everbridge products are trademarks of Everbridge, Inc. in the USA and other countries. All other product or company names mentioned are the property of their respective owners.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210809005689/en/
Everbridge:
Jeff Young
Media Relations
[email protected]
781-859-4116
Joshua Young
Investor Relations
[email protected]
781-236-3695
AP:
Lauren Easton
Global Director of Media Relations and Corporate Communications
[email protected]
212-621-7005
Patrick Maks
Media Relations Manager
[email protected]
212-621-7536
KEYWORDS: United States North America Massachusetts
INDUSTRY KEYWORDS: Data Management Communications Security Technology Software Networks Publishing
MEDIA:
| Photo |
 |
| Everbridge Teams with The Associated Press (AP) to Integrate Global News Reports into Market-Leading Critical Event Management (CEM) Platform (Graphic: Business Wire) |
| Logo |
 |