Ooma Adds Video Meetings to Ooma Office Pro, Making Collaboration Simpler and More Powerful
SUNNYVALE, Calif.–(BUSINESS WIRE)–Ooma, Inc., a smart communications platform for businesses and consumers, today announced availability of the Ooma Meetings video collaboration platform as part of its Ooma Office Pro business phone service.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20201118005162/en/
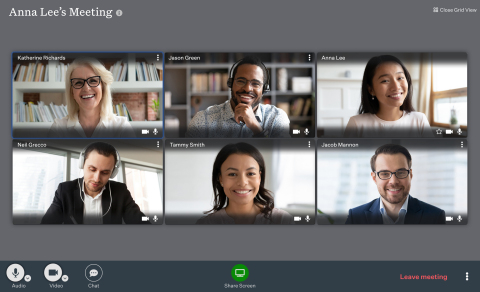
Ooma today announced availability of the Ooma Meetings video collaboration platform as part of its Ooma Office Pro business phone service. Unlike many other videoconferencing systems, Ooma Meetings (www.ooma.com/small-business-phone-systems/video-conferencing/) makes it easy for multiple users to share their screens simultaneously. One user can share a spreadsheet, for example, while another user shares a presentation slide and a third user shares a document. Participants can click on the shared screen of their choice to easily view it in the main window. (Graphic: Business Wire)
Ooma Office Pro users can manage and join video meetings through the existing Ooma Office desktop app, which turns any computer into a full-featured desk phone, also known as softphone. Ooma Meetings is also expected to be available soon through a mobile client for iOS and Android smart phones and tablets.
Unlike many other videoconferencing systems, Ooma Meetings (www.ooma.com/small-business-phone-systems/video-conferencing/) makes it easy for multiple users to share their screens simultaneously. One user can share a spreadsheet, for example, while another user shares a presentation slide and a third user shares a document. Participants can click on the shared screen of their choice to easily view it in the main window.
What’s more, for many users, Ooma Meetings requires no software installation. On desktop and laptop computers, video meetings can run directly inside Chrome, Safari or Edge browsers – users only need to click a web link to join. No app installation or browser extension is required. This removes a significant barrier for businesses when sending meeting invites to customers and partners who might be reluctant to install additional software. Users who aren’t near a computer have the option to dial into meetings by phone.
The clean Ooma Meetings interface is designed for intuitive collaboration. Additional features of Ooma Meetings include:
- Chat
- Host can mute an individual or all other participants
- One-time and recurring meetings
- Personal meeting ID
- “Meet now” function for ad hoc meetings
- Option to require a password to join a meeting
To see Ooma Meetings in action, watch a two-minute video at https://www.youtube.com/watch?v=dQMUb_sW4Bw.
“My architect clients are very impressed with Ooma Meetings because we can all share our screens,” said Peter A. Hugh of Hugh Lighting Design, an architectural lighting design firm in Oak Park, Ill. “One client said, as we were working together, ‘This is like passing notes across a table.’ Also, small businesses like mine are re-assessing value in the pandemic era and Ooma Office is now even more worthwhile for me because I don’t need to spend more for videoconferencing or manage yet another service.”
“More than ever, businesses need access to powerful communications tools such as video conferencing and screen sharing, both to support workers who work remotely as well to maintain a real sense of connection with colleagues, customers and partners,” said Dennis Peng, vice president of product management at Ooma. “Ooma Meetings is intended to empower businesses with the collaboration tools they need to thrive in this new ‘work from anywhere’ era.”
Ooma Meetings is available now to Ooma Office Pro (https://www.ooma.com/office/pro/) customers at no extra cost; the monthly fee for Ooma Office Pro remains a straightforward $24.95 per user, plus taxes and fees, with no contract required. Up to 25 users can join video meetings lasting up to two hours.
Current Ooma Office Pro users can activate Ooma Meetings by asking their account administrator to follow the instructions at https://support.ooma.com/office/ooma-meetings-overview/.
About Ooma, Inc.
Ooma (NYSE: OOMA) creates powerful connected experiences for businesses and consumers, delivered from its smart cloud-based SaaS platform. For businesses of all sizes, Ooma provides advanced voice and collaboration features that are flexible and scalable. For consumers, Ooma’s residential phone service provides PureVoice HD voice quality, advanced functionality and integration with mobile devices. Ooma’s innovative smart security solution delivers a range of wireless security sensors that make it easy for anyone to protect their home. Learn more at www.ooma.com or www.ooma.ca in Canada.
View source version on businesswire.com: https://www.businesswire.com/news/home/20201118005162/en/
MEDIA CONTACT:
Mike Langberg at Ooma
[email protected]
650-566-6693
INVESTOR CONTACT:
Matt Robison at Ooma
[email protected]
650-300-1480
KEYWORDS: California United States North America
INDUSTRY KEYWORDS: Technology VoIP Audio/Video Telecommunications Mobile/Wireless Software Internet
MEDIA:
| Photo |
 |
| Ooma today announced availability of the Ooma Meetings video collaboration platform as part of its Ooma Office Pro business phone service. Unlike many other videoconferencing systems, Ooma Meetings (www.ooma.com/small-business-phone-systems/video-conferencing/) makes it easy for multiple users to share their screens simultaneously. One user can share a spreadsheet, for example, while another user shares a presentation slide and a third user shares a document. Participants can click on the shared screen of their choice to easily view it in the main window. (Graphic: Business Wire) |
