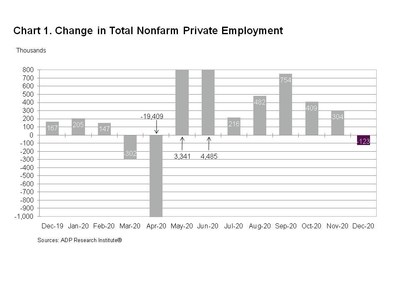
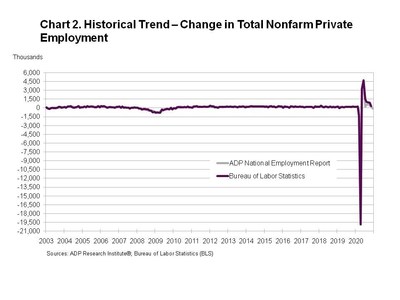
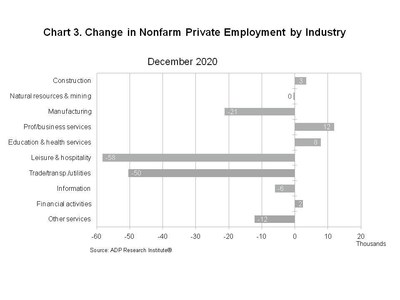
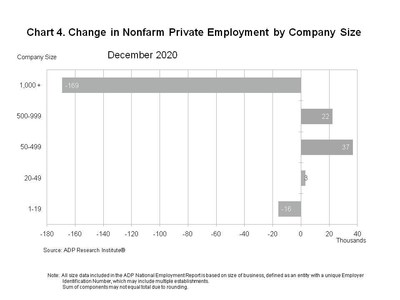
-
Company to Expand R&D Focus for Pipeline Growth to include Earlier Stage Programs and Validated Technology Platforms
-
Firdapse® 2020 Net Revenues Estimate of approximately $118 Million; about 16% Increase From 2019
-
Ended 2020 with Estimated $140 Million in Cash and Investments
-
Company Highlights Current Product Development Programs
CORAL GABLES, Fla., Jan. 06, 2021 (GLOBE NEWSWIRE) — Catalyst Pharmaceuticals, Inc. (Catalyst) (Nasdaq: CPRX), a commercial-stage biopharmaceutical company focused on developing and commercializing innovative therapies for people with rare debilitating, chronic neuromuscular and neurological diseases, today announced an expansion in its strategic priorities related to portfolio planning and research and development. Catalyst also provided preliminary net revenues and cash on hand for fiscal-year 2020, as well as an update on product development activities and current litigation.
“Our board and management team are aligned in the opportunity to recalibrate our strategic priorities for 2021 and for the long-term. We believe that we have the ability to leverage our solid balance sheet and strong cash flow, along with our expertise, in developing and commercializing additional medicines to treat rare and ultra-rare diseases. With this in mind, we intend to move to expand our product portfolio beyond amifampridine, with a focus on validated technology platforms as well as earlier stage programs in other rare disease therapeutic categories,” said Patrick J. McEnany, Catalyst Pharmaceuticals’ Chairman and CEO.
Mr. McEnany continued: “Additionally, I am pleased to report our preliminary estimate indicates that 2020 Firdapse® net revenues were approximately $118 million, an increase of $16 million from 2019. This reflects strong sales performance in an extremely difficult operating environment because of the COVID-19 pandemic. Lastly, we ended the year with approximately $140 million in cash and investments, which represents an increase of approximately $46 million compared to cash and investments at December 31, 2019, and we continue to have no funded debt.”
Strategic Plan for 2021 and Beyond
Catalyst’s Board of Directors recently approved an expansion in the company’s strategic focus to include acquiring or in-licensing innovative, validated technology platforms and earlier stage programs in other therapeutic categories outside of neuromuscular diseases. To accomplish these new priorities, Catalyst is prepared to invest more heavily in research and development, including acquiring earlier stage opportunities and validated innovative technology. Catalyst believes that this strategic expansion better positions the company to build out a broader more diversified portfolio of drug candidates that Catalyst expects will add greater value to the company over the near and long-term.
To spearhead this investment, Catalyst has commenced a national search for a key executive to manage this more progressive strategy. This will likely be an M.D. or Ph.D. with 15-20 years of relevant pharma experience and experience in innovated drug technology. This person will be responsible for portfolio planning and developing medicines from discovery through marketing authorizations, as well as strategic leadership across all R&D activities including, direct oversight of science and clinical research.
Year End 2020 Estimated Financial Results
Firdapse annual net revenues for 2020 are estimated at approximately $118 million, an increase of approximately 16% from 2019. Fiscal-year 2020 was a difficult period for patients and healthcare providers in many ways, including the ability to receive a definitive diagnosis of LEMS and providing access to therapies like Firdapse®. Catalyst believes that as COVID-19 vaccines are administered over the next few quarters, Catalyst will see a more robust uptake in new patient starts and revenues.
Catalyst also estimates that it ended 2020 with approximately $140 million in cash and investments, compared to $94.5 million at the end of 2019, up $46 million, or approximately 48%.
Year-End 2020 Status of Product Development Programs
Firdapse LA (Long-Acting)
The development of the long-acting formulation of amifampridine phosphate remains on track. A large number of candidate formulations were prepared, and three of the most promising formulations were evaluated in a pharmacokinetic (PK) study completed during the fourth quarter of 2020. The results from this first PK study will be used to inform the design and refinement of future product formulations in 2021, and additional PK work is also expected to be conducted during 2021. Catalyst has also completed a number of advisory board meetings with both patients and doctors in order to establish the optimum target characteristics of Firdapse LA that are desired by the LEMS patient community and treating physicians. Periodic updates on this program will be provided as it proceeds.
HNPP
A proof-of-concept study evaluating Firdapse® as a treatment for Hereditary Neuropathy with Liability to Pressure Palsies (HNPP) is scheduled to start in the first quarter of 2021. The scientific basis for considering this indication is that leakage of neuron potassium channels is observed in HNPP. Firdapse is a potassium channel blocker and may mitigate the pathological effects of the potassium channel leakage in HNPP patients.
HNPP is an autosomal-dominantly inherited peripheral nerve disease caused by a heterozygous deletion of PMP22 gene, leading to a reduction of PMP22 proteins by 35-50% of normal level, which results in disruption of the myelin sheaths of motor neuron axons. Patients with HNPP often present with focal sensory and motor deficits. The events may be triggered by mild mechanical compressions innocuous to healthy humans. In addition, HNPP patients are also afflicted by fatigue. Catalyst believes that HNPP affects about 6,000 patients in the Unites States.
It is hypothesized that functional demyelination in HNPP results in excessive outward current through internodal voltage-gated potassium channels. Application of a potassium channel blocker, like amifampridine, is expected to improve action potential propagation in HNPP, thereby alleviating sensory/motor deficits and fatigue in HNPP. This hypothesis is supported by results in a HNPP animal model with PMP22 heterozygous deletion (PMP22+/-).
MuSK-Myasthenia Gravis
Catalyst has concluded its detailed analysis of potential reasons why its MuSK-MG Phase 3 trial did not meet statistical significance on its endpoints. As reported previously, marked persistent clinical improvement was observed by patients and investigators during the initial dose-titration period of this trial and in the previous proof of concept trial. Catalyst also found that there was a large degree of symptom variability during the double-blind withdrawal period. Catalyst believes these sources of variability can be dealt with in a redesigned study that may better demonstrate the observed efficacy. Catalyst plans to present its hypotheses and a revised protocol to the FDA for discussion during the first half of 2021. However, there can be no assurance that FDA will accept the results of a single study of a different design as sufficient evidence for approval of the MuSK-MG indication. While Catalyst prepares for a meeting with the FDA, Catalyst will evaluate new clinical trial sites and discuss the new trial design with investigators. After the meeting with the FDA, Catalyst will determine whether or not to proceed with a new trial that incorporates the new trial design.
SMA-001
Catalyst’s exploratory study, SMA-001 (A Randomized Placebo Controlled Crossover Study to Evaluate the Safety and Efficacy of Amifampridine Phosphate in Ambulatory Patients with Spinal Muscular Atrophy (SMA) Type 3, met the primary endpoint of a statistically significant difference for the Hammersmith Functional Motor Scale Expanded (HFSME). Clinically, however, the effect was modest. The secondary endpoints were not statistically significant, although several individual quality of life measures demonstrated a positive statistically significant change. Key opinion leaders with whom Catalyst has spoken believed that Firdapse® needed to show a large clinically significant change if there was the possibility to affect disease progression through retrograde signaling from enhanced neuromuscular junction function. After considering all of these factors, Catalyst has concluded that the modest results exhibited in this study are unlikely to result in a sufficient modification of disease progression, and, particularly in light of the fact that there are now three approved disease modifying medications for SMA Type 3, Catalyst has decided not to pursue the SMA Type 3 indication further.
Kennedy’s disease
Previously, key opinion leaders had suggested that Firdapse may be effective in treating the symptoms of Kennedy’s disease (spinal and bulbar muscular atrophy). However, given the results of the SMA trial and the lack of a targeted mechanism of action for Firdapse in Kennedy’s disease, Catalyst has decided not to pursue this indication further.
Litigation Update
In Catalyst’s appeal of the U.S. District Court’s decision in its suit challenging the Food and Drug Administration’s interpretation of Firdapse’ s orphan drug exclusivity under the Orphan Drug Act, the briefs have been filed, and Catalyst is currently awaiting either a schedule for oral arguments or a decision from the 11th Circuit Court of Appeals. If oral arguments are scheduled, it is still anticipated that they will not occur for a few months.
Catalyst’s previously announced patent litigation against Jacobus Pharmaceuticals Inc. and PANTHERx, Jacobus’ sole specialty pharmacy provider for Ruzurgi®, is proceeding as expected. These types of cases, however, take a long time to develop through discovery and other pre-trial activities so no trial date is expected for the foreseeable future.
About Catalyst Pharmaceuticals
Catalyst Pharmaceuticals is a commercial-stage biopharmaceutical company focused on developing and commercializing innovative therapies for people with rare debilitating, chronic neuromuscular and neurological diseases, including Lambert-Eaton myasthenic syndrome (LEMS), anti-MuSK antibody positive myasthenia gravis (MuSK-MG) and other neurological and neuromuscular disorders. Catalyst’s new drug application for Firdapse® (amifampridine) 10 mg tablets for the treatment of adults with LEMS was approved in 2018 by the U.S. Food & Drug Administration (“FDA”), and Firdapse is now commercially available in the United States. Further, Canada’s national healthcare regulatory agency, Health Canada, recently approved the use of Firdapse® (amifampridine) for the treatment of patients in Canada with LEMS.
Firdapse is currently being evaluated in clinical trials for the treatment of MuSK-MG and has received Orphan Drug Designation from the FDA for myasthenia gravis.
Forward-Looking Statements
This press release contains forward-looking statements. Forward-looking statements involve known and unknown risks and uncertainties, which may cause Catalyst’s actual results in future periods to differ materially from forecasted results. A number of factors, including (i) whether Catalyst can retain a key executive with the qualifications and experience that Catalyst seeks to lead Catalyst’s efforts to source and develop new products and/or technologies, (ii) whether Catalyst’ planned efforts to source and acquire or in-license innovated, validated technology platforms and earlier stage programs in new therapeutic categories will be successful, (iii) whether Catalyst will continue to be profitable and cash flow positive in future periods, (iv) whether Catalyst can successfully expand the number of LEMS patients being treated with Firdapse
®
, (v) whether Catalyst’s plans for a long-acting amifampridine product, and its plans to further evaluate Firdapse
®
for the treatment of MuSK-MG and HNPP, will be successful, (vi) whether Catalyst’s appeal of the District Court’s decision in its lawsuit against the FDA to overturn the approval of Ruzurgi
®
will be successful, (vii) the scope of protection from competition provided by Catalyst’s recently granted patent, and (viii) those factors described in Catalyst’s Annual Report on Form 10-K for fiscal year 2019 and its other filings with the U.S. Securities and Exchange Commission (SEC), could adversely affect Catalyst. Copies of Catalyst’s filings with the SEC are available from the SEC, may be found on Catalyst’s website, or may be obtained upon request from Catalyst. Catalyst does not undertake any obligation to update the information contained herein, which speaks only as of this date.


![]() View original content to download multimedia:http://www.prnewswire.com/news-releases/adp-national-employment-report-private-sector-employment-decreased-by-123-000-jobs-in-december-301201960.html
View original content to download multimedia:http://www.prnewswire.com/news-releases/adp-national-employment-report-private-sector-employment-decreased-by-123-000-jobs-in-december-301201960.html