Study Investigates the Effects of Ventilatory Rescue Therapies on the Cerebral Oxygenation of COVID-19 Patients Using Masimo O3®
Study Stresses the Importance of Brain Monitoring During the Treatment of Patients with Severe COVID-19
NEUCHATEL, Switzerland–(BUSINESS WIRE)–Masimo (NASDAQ: MASI) today announced the results of a prospective, observational study published in Critical Care in which researchers in Genoa, Italy, evaluated the impact of a variety of rescue therapies on the systemic and cerebral oxygenation of mechanically ventilated COVID-19 patients suffering from acute respiratory distress syndrome (ARDS).1 To gauge the impact, the researchers used the Masimo Root® Patient Monitoring and Connectivity Platform with O3® Regional Oximetry, which uses near-infrared spectroscopy (NIRS) to enable monitoring of tissue oxygen saturation (rSO2) in the region of interest, such as the brain.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20210405005487/en/
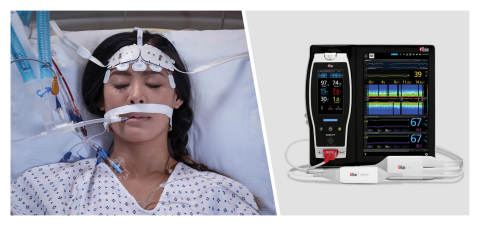
Masimo Root® with O3® Regional Oximetry and SedLine® Brain Function Monitoring (Photo: Business Wire)
Dr. Chiara Robba and colleagues noted that “neurological complications are common in mechanically ventilated critically ill patients with COVID-19 and may lead to impaired cerebral hemodynamics,” and further, that respiratory rescue therapies “may have detrimental effects on brain physiology.” Observing, however, that there is currently little data available regarding the effect of rescue therapies on these patients’ brains, and in particular on cerebral oxygenation, the researchers sought to assess the impact of different ventilatory rescue therapies on the brain to help guide clinicians in choosing the most appropriate therapies for their COVID-19 patients.
The rescue therapies studied were recruitment maneuvers (RMs), prone positioning (PP), inhaled nitric oxide (iNO), and extracorporeal carbon dioxide removal (ECCO2R). To assess impact, the researchers measured (before and after the application of each method) arterial oxygen saturation (SpO2), partial pressure of oxygen (PaO2), partial pressure of carbon dioxide (PaCO2), and cerebral oxygen saturation (rSO2). rSO2 was obtained using Masimo Root with O3, which also allowed them to observe several additional parameters unique to Masimo O3: ΔO2Hb, which monitors relative changes in the oxygenated hemoglobin component of rSO2; ΔHHb, which monitors relative changes in the deoxygenated hemoglobin component of rSO2; and ΔcHb, which monitors relative changes in total cerebral hemoglobin or blood volume. As a secondary aim, the researchers sought to evaluate the correlation between systemic and cerebral oxygenation.
The researchers found that the four rescue therapies had varied impact on cerebral oxygenation and the other measured parameters, noting in particular that after RMs, while there was no significant change in PaO2 or PaCO2, there was a significant decrease in rSO2. After PP and after iNO therapies, both PaO2 and rSO2 increased; ΔcHb also increased, corresponding to increased cerebral blood volume. After ECCO2R, both PaO2 and rSO2 decreased.
The researchers concluded, “Rescue therapies exert specific pathophysiological mechanisms, resulting in different effects on systemic and cerebral oxygenation in critically ill COVID-19 patients with ARDS. … The choice of rescue strategy to be adopted should take into account both lung and brain needs.”
They also noted, “To our knowledge, this is the first study investigating the early effects of rescue therapies on systemic and cerebral oxygenation and their correlation in critically ill patients with COVID-19-associated ARDS. The use of multimodal neuromonitoring, including new indices such as ΔHHbi + ΔO2Hbi, enabled us to better investigate the specific consequences of each ventilatory rescue strategy for brain and lung function. This is particularly important, especially in the early phases after rescue therapies application, when most of the effects on cerebral physiology are mainly acting.”
Dr. Robba and study co-author Dr. Basil Matta, Senior Medical Director at Masimo, commented, “The ability to observe relative changes in oxygenated, deoxygenated, and total hemoglobin with O3’s delta indices provided us with better insight into why brain saturations change as a result of interventions, and allowed us to better understand the interactions between systemic and cerebral hemodynamics. For example, we saw that turning patients prone resulted in improved systemic and cerebral oxygenation, whereas the lung recruitment maneuver did not improve systemic oxygenation, and even had an adverse effect by reducing brain oxygen saturation.”
They continued, “Above all, the main objective of improving the oxygen content of the blood is to deliver oxygen to vital organs, the most important of which is the brain. Masimo O3 provides the clinician with the ability to assess the impact of any medical intervention aimed at improving oxygenation. O3’s hemoglobin indices were critical to our understanding of the effects of our interventions on the brain. Without such a monitor, we are at best guessing, and in danger of flying blind. As we continue to seek to improve care and outcomes for patients with severe COVID-19, any tool that helps us better understand the impact of different medical interventions is most welcome.”
@Masimo | #Masimo
About Masimo
Masimo (NASDAQ: MASI) is a global medical technology company that develops and produces a wide array of industry-leading monitoring technologies, including innovative measurements, sensors, patient monitors, and automation and connectivity solutions. Our mission is to improve patient outcomes and reduce the cost of care. Masimo SET® Measure-through Motion and Low Perfusion™ pulse oximetry, introduced in 1995, has been shown in over 100 independent and objective studies to outperform other pulse oximetry technologies.2 Masimo SET® has also been shown to help clinicians reduce severe retinopathy of prematurity in neonates,3 improve CCHD screening in newborns,4 and, when used for continuous monitoring with Masimo Patient SafetyNet™ in post-surgical wards, reduce rapid response team activations, ICU transfers, and costs.5-8 Masimo SET® is estimated to be used on more than 200 million patients in leading hospitals and other healthcare settings around the world,9 and is the primary pulse oximetry at 9 of the top 10 hospitals according to the 2020-21 U.S. News and World Report Best Hospitals Honor Roll.10 Masimo continues to refine SET® and in 2018, announced that SpO2 accuracy on RD SET® sensors during conditions of motion has been significantly improved, providing clinicians with even greater confidence that the SpO2 values they rely on accurately reflect a patient’s physiological status. In 2005, Masimo introduced rainbow® Pulse CO-Oximetry technology, allowing noninvasive and continuous monitoring of blood constituents that previously could only be measured invasively, including total hemoglobin (SpHb®), oxygen content (SpOC™), carboxyhemoglobin (SpCO®), methemoglobin (SpMet®), Pleth Variability Index (PVi®), RPVi™ (rainbow® PVi), and Oxygen Reserve Index (ORi™). In 2013, Masimo introduced the Root® Patient Monitoring and Connectivity Platform, built from the ground up to be as flexible and expandable as possible to facilitate the addition of other Masimo and third-party monitoring technologies; key Masimo additions include Next Generation SedLine® Brain Function Monitoring, O3® Regional Oximetry, and ISA™ Capnography with NomoLine® sampling lines. Masimo’s family of continuous and spot-check monitoring Pulse CO-Oximeters® includes devices designed for use in a variety of clinical and non-clinical scenarios, including tetherless, wearable technology, such as Radius-7® and Radius PPG™, portable devices like Rad-67™, fingertip pulse oximeters like MightySat® Rx, and devices available for use both in the hospital and at home, such as Rad-97®. Masimo hospital automation and connectivity solutions are centered around the Masimo Hospital Automation™ platform, and include Iris® Gateway, iSirona™, Patient SafetyNet, Replica™, Halo ION™, UniView™, UniView :60™, and Masimo SafetyNet™. Additional information about Masimo and its products may be found at www.masimo.com. Published clinical studies on Masimo products can be found at www.masimo.com/evidence/featured-studies/feature/.
ORi and RPVi have not received FDA 510(k) clearance and are not available for sale in the United States. The use of the trademark Patient SafetyNet is under license from University HealthSystem Consortium.
References
-
Robba C, Ball L, Battaglini D, Cardim D, Moncalvo E, Brunetti I, Bassetti M, Giacobbe D, Vena A, Patroniti N, Rocco P, Matta B, Pelosi P. Early effects of ventilatory rescue therapies on systemic and cerebral oxygenation in mechanically ventilated COVID-19 patients with acute respiratory distress syndrome: a prospective observational study. Crit Care (2021)25:111. DOI: https://doi.org/10.1186/s13054-021-03537-1.
-
Published clinical studies on pulse oximetry and the benefits of Masimo SET® can be found on our website at http://www.masimo.com. Comparative studies include independent and objective studies which are comprised of abstracts presented at scientific meetings and peer-reviewed journal articles.
-
Castillo A et al. Prevention of Retinopathy of Prematurity in Preterm Infants through Changes in Clinical Practice and SpO2 Technology. Acta Paediatr. 2011 Feb;100(2):188-92.
-
de-Wahl Granelli A et al. Impact of pulse oximetry screening on the detection of duct dependent congenital heart disease: a Swedish prospective screening study in 39,821 newborns. BMJ. 2009;Jan 8;338.
-
Taenzer A et al. Impact of pulse oximetry surveillance on rescue events and intensive care unit transfers: a before-and-after concurrence study. Anesthesiology. 2010:112(2):282-287.
-
Taenzer A et al. Postoperative Monitoring – The Dartmouth Experience. Anesthesia Patient Safety Foundation Newsletter. Spring-Summer 2012.
-
McGrath S et al. Surveillance Monitoring Management for General Care Units: Strategy, Design, and Implementation. The Joint Commission Journal on Quality and Patient Safety. 2016 Jul;42(7):293-302.
-
McGrath S et al. Inpatient Respiratory Arrest Associated With Sedative and Analgesic Medications: Impact of Continuous Monitoring on Patient Mortality and Severe Morbidity. J Patient Saf. 2020 14 Mar. DOI: 10.1097/PTS.0000000000000696.
-
Estimate: Masimo data on file.
- http://health.usnews.com/health-care/best-hospitals/articles/best-hospitals-honor-roll-and-overview.
Forward-Looking Statements
This press release includes forward-looking statements as defined in Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, in connection with the Private Securities Litigation Reform Act of 1995. These forward-looking statements include, among others, statements regarding the potential effectiveness of Root® with O3®. These forward-looking statements are based on current expectations about future events affecting us and are subject to risks and uncertainties, all of which are difficult to predict and many of which are beyond our control and could cause our actual results to differ materially and adversely from those expressed in our forward-looking statements as a result of various risk factors, including, but not limited to: risks related to our assumptions regarding the repeatability of clinical results; risks related to our belief that Masimo’s unique noninvasive measurement technologies, including Root with O3, contribute to positive clinical outcomes and patient safety; risks related to our belief that Masimo noninvasive medical breakthroughs provide cost-effective solutions and unique advantages; risks related to COVID-19; as well as other factors discussed in the “Risk Factors” section of our most recent reports filed with the Securities and Exchange Commission (“SEC”), which may be obtained for free at the SEC’s website at www.sec.gov. Although we believe that the expectations reflected in our forward-looking statements are reasonable, we do not know whether our expectations will prove correct. All forward-looking statements included in this press release are expressly qualified in their entirety by the foregoing cautionary statements. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of today’s date. We do not undertake any obligation to update, amend or clarify these statements or the “Risk Factors” contained in our most recent reports filed with the SEC, whether as a result of new information, future events or otherwise, except as may be required under the applicable securities laws.

View source version on businesswire.com: https://www.businesswire.com/news/home/20210405005487/en/
Masimo
Evan Lamb
949-396-3376
[email protected]
KEYWORDS: Europe Switzerland United States North America California
INDUSTRY KEYWORDS: Medical Devices Infectious Diseases Hospitals Health Medical Supplies
MEDIA:
| Logo |
 |
|
| Photo |
 |
| Masimo Root® with O3® Regional Oximetry and SedLine® Brain Function Monitoring (Photo: Business Wire) |






