PR Newswire
VANCOUVER, BC, April 7, 2021 /PRNewswire/ – New Found Gold Corp. (“New Found” or the “Company“) (TSXV: NFG) (OTC: NFGFF) is pleased to announce assay results from an additional ten holes drilled at the Keats Zone (“Keats“). These holes were drilled as part of the Company’s ongoing 200,000m diamond drill program at its 100%-owned Queensway Project (“Queensway“), located on the Trans-Canada Highway 15 km west of Gander, Newfoundland.
Highlights
- Highlights include:
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
*Note that the host structures are interpreted to be steeply dipping and true widths are estimated to be 70% to 80% of reported intervals. Intervals are calculated at a 1 g/t Au cut-off grade; grades have not been capped in the averaging. |
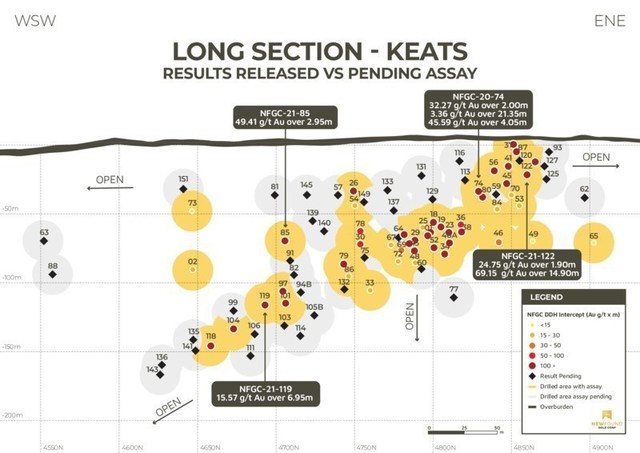
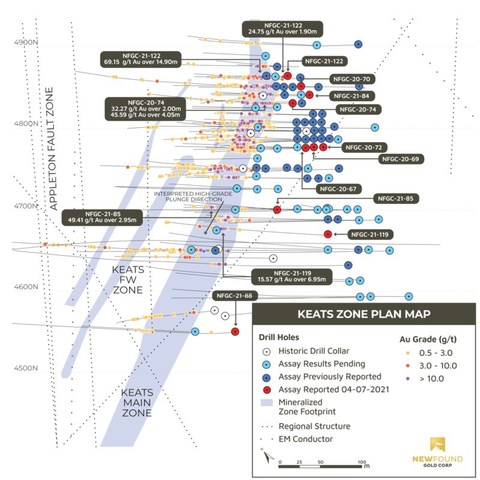
- Multiple intervals encountered in drill holes NFGC-21-74 and NFGC-21-122 provide further confirmation of high-grade, near surface gold mineralization at the north end of Keats. These results include 45.6g/t Au over 4.05m in hole NFGC-21-74 and 69.2g/t Au over 14.9m in hole NFGC-21-122 (Figure 1).
- The intervals of 49.4 g/t Au over 2.95m in drill hole NFGC-21-85 and 15.6 g/t Au over 6.95m NFGC 21-119 provide further confirmation of the continuity of the Keats high grade zone in the down-plunge direction (Figure 1).
Greg Matheson, P.Geo., COO of New Found, stated: “Similar to the results the Company disclosed on March 29, 2021, these results continue to infill between our deepest interval disclosed to date (61.8 g/t Au over 13.65m in NFGC-21-118) and the near-surface, high-grade results up-plunge (see Figure 1). Current drilling at Keats is focused on extending the high-grade zone down plunge, providing further definition of the near surface, high-grade mineralization, and testing additional zones of high-grade mineralization including in footwall veining.“
Highlight Keats Main Zone drill intervals for reference
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|||||||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
|
|
|
|||
|
|
|
|
|
|
|
|
|
|
Drillhole Details
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 2: Summary of results reported in this release.
|
*Note that the host structures are interpreted to be steeply dipping and true widths are estimated to be 70% to 80% of reported intervals. Intervals are calculated at a 1 g/t Au cut-off grade; grades have not been capped in the averaging. |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Table 3: Location details of drill holes reported on in this release.
Sampling, Sub-sampling and Laboratory
True widths of the intercepts reported in this press release have yet to be determined but are estimated to be 70% to 80% of reported core lengths. Assays are uncut, and calculated intervals are reported over a minimum length of 2 meters using a lower cut-off of 1.0 g/t Au. All HQ split core assays reported were obtained by either complete sample metallic screen/fire assay or standard 30-gram fire-assaying with ICP finish at ALS Minerals in Vancouver, British Columbia, or by entire sample screened metallic screen fire assay at Eastern Analytical in Springdale, Newfoundland. The complete sample metallic screen assay method is selected by the geologist when samples contain coarse gold or any samples displaying gold initial fire assay values greater than 1.0 g/t Au. Drill program design, Quality Assurance/Quality Control and interpretation of results is performed by qualified persons employing a Quality Assurance/Quality Control program consistent with National Instrument 43-101 and industry best practices. Standards and blanks are included with every 20 samples for Quality Assurance/Quality Control purposes by the Company as well as the lab. Approximately 5% of sample pulps are sent to secondary laboratories for check assays.
Qualified Person
The technical content disclosed in this press release was reviewed and approved by Greg Matheson, P. Geo., Chief Operating Officer, and a Qualified Person as defined under National Instrument 43-101. Mr. Matheson consents to the publication of this news release dated April 7, 2021 by New Found. Mr. Matheson certifies that this news release fairly and accurately represents the information for which he is responsible.
About New Found Gold Corp.
New Found holds a 100% interest in the Queensway Project, located 15 km west of Gander, Newfoundland, and just 18 km from Gander International Airport. The project is intersected by the Trans-Canada Highway and has logging roads crosscutting the project, high voltage electric power lines running through the project area, and easy access to a highly skilled workforce. The Company is currently undertaking a 200,000-m drill program at Queensway, with a planned increase from the current six drill rigs to eight drill rigs in Q1, 2021. New Found currently has working capital of approximately $64 million. On closing of the $15 million flow through financing announced March 18, 2021 the Company would have an estimated $79 million of working capital.
Please see the Company’s website at www.newfoundgold.ca and the Company’s SEDAR profile at www.sedar.com.
Acknowledgements
New Found acknowledges the financial support of the Junior Exploration Assistance Program, Department of Natural Resources, Government of Newfoundland and Labrador.
Contact
To contact the Company, please visit the Company’s website, www.newfoundgold.ca and make your request through our investor inquiry form. Our management has a pledge to be in touch with any investor inquiries within 24 hours.
New Found Gold Corp.
Per: “Craig Roberts”
Craig Roberts, P.Eng., Chief Executive Officer
Email: [email protected]
Phone: (604) 562 9664
Neither the TSX Venture Exchange nor its Regulation Services Provider (as that term is defined in the policies of the TSX Venture Exchange) accepts responsibility for the adequacy or accuracy of this release.
Forward-Looking Statement Cautions
This press release contains certain “forward-looking statements” within the meaning of Canadian securities legislation, relating to further the exploration and drilling
on the Company’s Queensway gold project in Newfoundland
. Although the Company believes that such statements are reasonable, it can give no assurance that such expectations will prove to be correct. Forward-looking statements are statements that are not historical facts; they are generally, but not always, identified by the words “expects,” “plans,” “anticipates,” “believes,” “intends,” “estimates,” “projects,” “aims,” “potential,” “goal,” “objective,” “prospective,” and similar expressions, or that events or conditions “will,” “would,” “may,” “can,” “could” or “should” occur, or are those statements, which, by their nature, refer to future events. The Company cautions that forward-looking statements are based on the beliefs, estimates and opinions of the Company’s management on the date the statements are made, and they involve a number of risks and uncertainties. Consequently, there can be no assurances that such statements will prove to be accurate and actual results and future events could differ materially from those anticipated in such statements. Except to the extent required by applicable securities laws and the policies of the TSX Venture Exchange, the Company undertakes no obligation to update these forward-looking statements if management’s beliefs, estimates or opinions, or other factors, should change. Factors that could cause future results to differ materially from those anticipated in these forward-looking statements include risks associated possible accidents and other risks associated with mineral exploration operations, the risk that the Company will encounter unanticipated geological factors, the possibility that the Company may not be able to secure permitting and other governmental clearances necessary to carry out the Company’s exploration plans, the risk that the Company will not be able to raise sufficient funds to carry out its business plans, and the risk of political uncertainties and regulatory or legal changes that might interfere with the Company’s business and prospects. The reader is urged to refer to the Company’s reports, publicly available through the Canadian Securities Administrators’ System for Electronic Document Analysis and Retrieval (SEDAR) at www.sedar.com for a more complete discussion of such risk factors and their potential effects.
![]() View original content to download multimedia:http://www.prnewswire.com/news-releases/keats-infill-drilling-returns-106-5-gt-au-over-9-15m-and-45-6-gt-au-over-4-05m-301263942.html
View original content to download multimedia:http://www.prnewswire.com/news-releases/keats-infill-drilling-returns-106-5-gt-au-over-9-15m-and-45-6-gt-au-over-4-05m-301263942.html
SOURCE New Found Gold Corp.